2020, June the 26th
Virus émergents et nouvelles maladies, conférence INSERM / Sciences et avenir.
Covid-19, ebola, zika, dengue, chikungunya...Depuis une vingtaine d’années, nous assistons à une résurgence de maladies virales. Les virus qui en sont à l’origine sont-ils plus virulents que les virus d’antan ? Que faut-il savoir à leur sujet ? Quels défis sont à relever ? Pour répondre à ces questions, l’Inserm a interrogé, en partenariat avec Sciences et Avenir, trois spécialistes des virus lors d’un webinar très instructif à réécouter.


2020, June the 8th
Cynbiose et le laboratoire académique VirPath s’associent dans un projet collaboratif visant à développer de nouveaux modèles précliniques pour l’évaluation de candidats thérapeutiques et prophylactiques contre le SARS-CoV-2 et ainsi en accélérer l’entrée en phases cliniques.
Coordonné par le pôle de compétivité Lyonbiopôle, en collaboration avec Vetagro Sup, et soutenu par la Région Auvergne-Rhône-Alpes, ce projet innovant et d'intérêt stratégique en santé illustre la capacité des acteurs académiques et industriels de notre écosystème régional à se mobiliser rapidement et travailler en synergie dans la lutte contre le COVID-19
Le projet COVIBIOSE s’inscrit dans une logique de partenariat à forte valeur ajoutée et de mise en œuvre de programme de recherche translationnelle à visées clinique et industrielle entre acteurs publics et privés, renforçant ainsi le positionnement national et international de notre pôle de compétitivité et de notre région Auvergne-Rhône-Alpes dans le domaine de la santé.
2020, June the 5th
Notre spin-off Vaxxel fait partie des 14 start-up innovantes françaises sélectionnées pour participer au programme Deeptech North America (NETVA) porté par le ministère de l’Europe et des Affaires Etrangères en partenariat avec le Ministère de l’Enseignement supérieur, de la recherche et de l’innovation et Bpifrance. Vaxxel bénéficiera ainsi de l’accompagnement du service pour la Science et la Technologie de l’Ambassade de France aux Etats Unis dans sa démarche d’internationalisation et de développement de son candidat vaccin vivant atténué contre les infections à pneumovirus.
2020, May the 25th
Unlike our previous observations for remdesivir, the antiviral activity of hydroxychloroquine in Vero E6 cells did not translate to our predictive human reconstituted respiratory epithelium, with doses of 1 μM or 10 μM failing to reduce significantly SARS-CoV-2 apical viral titres at 48 hours post-infection. Hydroxychloroquine also did not protect the epithelial integrity during infection, as evidenced by transepithelial electrical resistance (TEER) values comparable to untreated cells and significantly lower than those of the mock-infected controls.
Accordingly, in macaques, neither hydroxychloroquine nor hydroxychloroquine+azithromycin showed a significant effect on the viral load levels in any of the tested compartments. When the drug was used as a pre-exposure prophylaxis, hydroxychloroquine did not confer protection against acquisition of infection.
Altogether, results do not support the use of hydroxychloroquine, either alone or in combination with azithromycin, as an antiviral treatment for COVID-19 in humans.

This study was performed in close collaboration with Université Paris-Saclay, Inserm, CEA, IMVA-HB/IDMIT; Université de Paris, INSERM, IAME; Unité de Génétique Moléculaire des Virus à ARN, GMVR : Institut Pasteur, UMR CNRS 3569; Aix-Marseille Univ, APHM, Unité des Virus Emergents (UVE) IRD 190, INSERM 1207 and Unité des Virus Emergents, UVE: Aix Marseille Univ, IRD 190, INSERM 1207, IHU Méditerranée Infection
2020, May the 25th
Our last preprint on the Respiratory Syncytial Virus-induced immune signature of infection is now available.
Human Respiratory Syncytial Virus (HRSV) constitutes one the main causes of respiratory infection in neonates and infants worldwid. Our transcriptome analysis of nasal wash samples from HRSV infected children reveals a characteristic immune signature as a direct reflect of virus pathogenesis and highlights several putative biomarkers of interest.
This study was performed in close collaboration with Dr Guy Boivin's laboratory (CHU Québec, Université Laval)







2020, May the 22nd
Superinfections associated with COVID-19 include invasive aspergillosis. To better understand the biological impact of such superinfection, we have set up a SARS-CoV-2 Aspergillus superinfection model using our predictive reconstituted human respiratory epithelium in collaboration with Signia Therapeutics. Transcriptional profiling of superinfection reveals a type I/type III IFN imbalance and an induction of several monocyte and neutrophil associated chemokines that could contribute to a better understanding of the COVID-19 severe forms and the development of new therapies.
2020, May the 19th
Notre programme de recherche à visée thérapeutique contre le SARS-CoV-2 a reçu une subvention des Instituts de recherche en santé du Canada | Canadian Institutes of Health Research (IRSC).
Cet important financement va contribuer à l’accélération de nos projets de recherche menés en partenariat avec le laboratoire du Dr. Guy Boivin (Centre de recherche du CHU de Québec-Université Laval) et SIGNIA THERAPEUTICS. Basés sur une stratégie unique de repositionnement de médicaments déjà sur le marché pour de nouvelles indications thérapeutiques antivirales, nos programmes de R&D intègrent également l'évaluation dans plusieurs modèles précliniques prédictifs des composés candidats anti-SRAS-CoV-2 dans le but d'accélérer leur transfert en clinique.
2020, May the 14th
This study shows that influenza A virus infection durably impacts on the host’s systemic energy metabolism, and promotes depot-specific metabolic reprogramming of fat tissues, in mice.

2020, May the 12th
VirPath FR1660223 patent granted in France - Antiviral composition for the treatment of infections linked to coronaviruses.
VirPath announces the grant of its patent for the repurposing of marketed drugs as new antivirals against coronaviruses by the French Patent Office. An international patent application (WO 2018/073549 A1) and national phases are advanced. Licensed to SIGNIA THERAPEUTICS, this patent expands the company’s portfolio and highlights the strength of our strategy and SIGNATURA® platform for drug identification and repurposing as innovative host directed inhibitors of respiratory virus infections.


2020, May the 6th
Cela couronne avec succès les travaux de recherche et développement que nous avons menés depuis plusieurs années en partenariat avec les équipes du Pr Guy Boivin (Centre de recherche du CHU de Québec-Université Laval) et de la société de biotechnologie Transgene. Nos programmes de recherche qui ont conduit au développement de la plateforme vaccinale Metavac® et de son procédé de production basé sur la lignée cellulaire aviaire DuckCelt®-T17, aujourd’hui licenciées à la startup #Vaxxel, ont été soutenus activement par les filiales de l'Université Claude Bernard Lyon 1 (Lyon Ingnierie Projet et #EZUS Lyon) et financés par l'ANR (Agence nationale de la recherche), la Région Auvergne-Rhône-Alpes et la SATT PULSALYS. Vaxxel a été lauréate du concours i-Lab en 2019, organisé par le Ministère en charge de l'enseignement supérieur et de la recherche (France) en partenariat avec Bpifrance et a été labélisée FrenchTech seed en 2019.


2020, May the 4th
Naproxen binding to the nucleoprotein of SARS-CoV2 was predicted by molecular modelling. In VeroE6 cells and reconstituted human airway epithelium models of SARS-CoV-2 infection, naproxen inhibits viral replication and protects the bronchial epithelium against SARS-CoV-2 induced-damage. These results are issued from a fruitful collaboration with Sorbonne University, INSERM UMR S938 Centre de Recherche Saint-Antoine, UMR 8226 CNRS and APHP.
The benefit of anti-inflammatory and antiviral naproxen addition in the standard treatment of Critically Ill Patients Hospitalized for COVID-19 Infection is currently tested in the ENACOVID clinical trial (APHP).
2020, April the 28th
Therapeutic repurposing is about finding new therapeutic indications for treatments that are well-known, safe and already available. Within the context of the COVID-19 pandemic, a number of teams are testing molecules used for other diseases on SARS-CoV-2, in order to evaluate their effects. The team of Inserm researcher Manuel Rosa-Calatrava at the International Center for Research in Infectious Diseases (Inserm/Université Claude-Bernard Lyon 1/CNRS/ENS Lyon) is working on the development and validation of a strategy to repurpose drugs for new antiviral therapeutic indications. Having already repurposed in vitro two molecules available on the market for respiratory viruses, including MERS-CoV, the researchers are now testing them against SARS-CoV-2 on cell lines and on an ex vivo model using reconstituted human respiratory epithelium.
2020, April the 22nd
Notre équipe est partenaire de trois projets sélectionnés dans le cadre de l’appel Flash Covid-19 de l’ANR.
2020, April the 20th
Observation intracellulaire d’épithélium respiratoire humain reconstitué MucilAir™ infecté par le SARS-CoV-2.
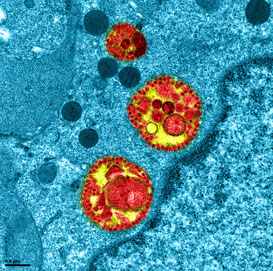
Des vésicules cytoplasmiques très caractéristiques contenant de larges accumulations de matériel viral, des membranes et de nombreux virus en assemblage, sont observées en région périnucléaire de cellule épithéliale ciliée. Ces structures viro-induites typiques du cycle réplicatif des coronavirus, illustrent le réarrangement des compartiments cellulaires (réticulum et appareil de Golgi) permettant le transport des virus vers la membrane cellulaire et leur libération au pôle apical de l’épithélium. Cette image a été obtenue par microscopie électronique à transmission sur la plateforme d’imagerie l'Université Claude Bernard Lyon 1.
Crédits photos: Manuel Rosa-Calatrava, INSERM ; Olivier Terrier, CNRS ; Andrés Pizzorno, SIGNIA THERAPEUTICS ; Elisabeth Errazuriz-Cerda UCBL1 CIQLE. hashtag#VirPath (CIRI U1111 INSERM - UMR 5308 CNRS - ENS Lyon - UCBL1). Colorisé par Noa Rosa C.
2020, April the 8th
Interview of Dr Manuel Rosa-Caltrava, by David Gossart (Tribune de Lyon) about the solutions to fight the COVID-19 pandemic.
2020, April the 6th
Article of "Lyon Capitale" about laboratories in Lyon and COVID-19.